
Teleflex is the home of Arrow®, Deknatel®, LMA®, Pilling®, QuikClot®, Rusch®, UroLift®, and Weck® – trusted brands united by a common sense of purpose.įorward-Looking StatementsAny statements contained in this press release that do not describe historical facts may constitute forward-looking statements. Teleflex employees worldwide are united in the understanding that what we do every day makes a difference. Our portfolio is diverse, with solutions in the fields of vascular access, interventional cardiology and radiology, anesthesia, emergency medicine, surgical, urology and respiratory care. We apply purpose driven innovation – a relentless pursuit of identifying unmet clinical needs – to benefit patients and healthcare providers. Both catheters will be on display as part of Teleflex’s hands-on workshops during CTO Plus: Complex CTO & Higher Risk PCI, February 23-24 at the New York Marriott Marquis in Times Square.Ībout Teleflex IncorporatedTeleflex is a global provider of medical technologies designed to improve the health and quality of people’s lives. The GuideLiner® Coast™ Catheter is currently in a limited market release phase, with full market release anticipated later this year. 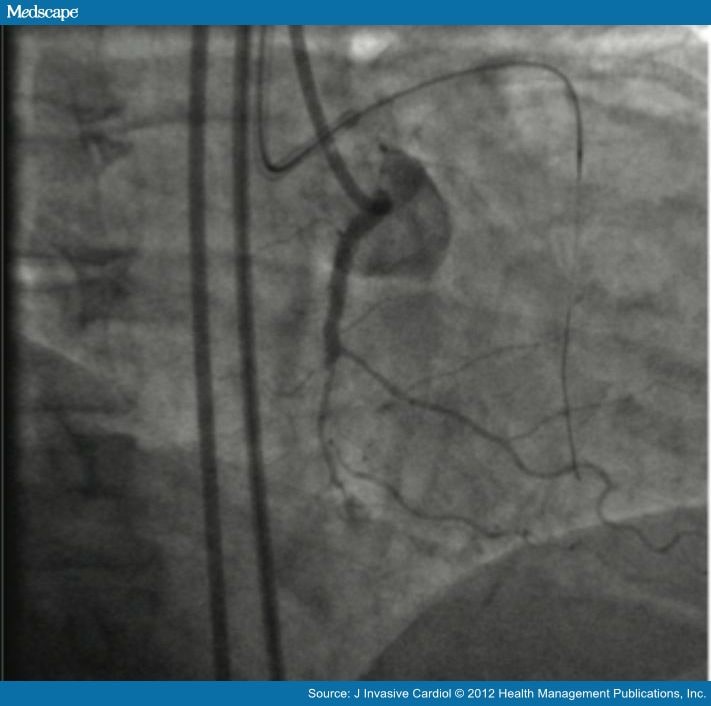
“The Triumph™ Catheter and the GuideLiner® Coast™ Catheter reflect our focus on providing physicians with new choices and technologies engineered to address unmet clinical needs, personal preference and technique, and the challenges of complex anatomy.” “Teleflex is committed to providing more options in the cath lab to help interventionalists address the specific needs of their patients,” said Scott Holstine, President and General Manager of Teleflex’s Interventional business unit. The GuideLiner® Coast™ Catheter adds a hydrophilic option to Teleflex’s market-leading guide extension portfolio-anchored by the flagship GuideLiner® V3 Catheter-enabling physicians to select the level of deliverability and backup support needed for specific cases.1 Earlier this year, the GuideLiner® Coast™ Catheter was first used in a clinical procedure at the UW Medicine Heart Institute in Seattle, Washington. “The Triumph™ Catheter’s nitinol ports and cage bring navigation and control to a new level, and I am excited that Teleflex will enable interventional cardiologists to readily access this important technology.” “In complex cases, the ability to better navigate tortuous anatomy has long been a challenge,” said Dr. Designed in collaboration with Bill Nicholson, MD, Director of Interventional Cardiology at Emory Healthcare, Atlanta, GA, the Triumph™ Catheter features a unique design with six wire exit ports for precise wire advancement and clear visualization. The Triumph™ Catheter is the latest addition to the Teleflex family of catheters, which includes the GuideLiner® V3 Catheter, TrapLiner® Catheter, and Turnpike® Catheters. 
Food and Drug Administration (FDA) 510(k) clearance of the Triumph™ Catheter, and the first clinical use of GuideLiner® Coast™ Catheter. Teleflex Incorporated (NYSE: TFX), a leading global provider of medical technologies, today announced key milestones in the release of two new catheters: the U.S. Create New Watchlist Create Create a new holdings portfolio Add Create + Add another position Close
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
